About Us
The Center provides fish health services to Federally funded national fish hatcheries within California, Nevada, and southern Oregon. In addition, the center works in cooperation with other Federal, state and Tribal agencies in surveying, sampling and analyzing fish populations.
What We Do
The CA-NV Fish Health Center is a BSL-2 laboratory providing aquatic animal health services, including inspections, diagnostics, and monitoring related to both hatchery and wild fish populations. The Center also addresses emerging pathogen issues through the Wild Fish Heath Survey and applied research.
Bacteriology:
Bacteria is one of the primary causes of disease in fish. The detection, prevention, and control of infectious diseases for fish remains one of the most important considerations for their successful culture. Worldwide, approximately 35 species of bacteria have been isolated from or associated with diseased fish. Of these, 12 species of bacteria are considered major pathogens of fish, and five are considered obligate pathogens (only causes disease in fish).
Growth and isolation of bacteria on appropriate media is the standard method used for diagnostic screening, monitoring of stocks, and fish health inspections for bacterial pathogens. Initial isolation and biochemical tests are preformed to identify the bacteria. The bacterial culture can also be tested against various antibiotics for effective treatment of some diseases. Bacteriological testing, from collection to positive identification, usually takes 3-4 days to complete.
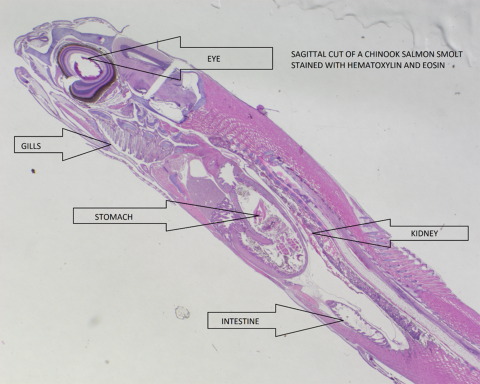
Histology:
Histology is an important field of fish health that can often detect subtle conditions or early signs of disease not easily recognized on gross examination. Changes at the cellular level and in the function of key organs are the first indications of mal-adjustment to stressors that may eventually lead to poor health or disease. Histology in the wild fish survey supplements general observations and pathogen screening of wild fish populations. Special projects such as water quality and contaminant studies can be augmented with histology to provide better insight into the environmental and/or physiological demands presented to fish in their natural environment.
Virology:
Viruses are obligate, submicroscopic, intracellular parasites that depend upon living cells for their reproduction and cannot propagate in the absence of living cells. All known viruses are infective agents, are often host specific, and in some cases have a high specificity for certain tissues. The isolation and identification of viruses is an elaborate process and requires sophisticated equipment. New methods continue to be developed, but there are several standard techniques used for laboratory diagnosis of viral infections. Many methods utilize in vitro cell culture (tissue culture) systems which are susceptible to cytopathological effects (CPE) from the virus and molecular techniques for confirmation.
As there are no effective treatments for viral infections of hatchery fish; regular monitoring of juvenile and adults, sanitation and disinfection are the only practical methods of control.
Projects and Research
Applied Research:
The Center conducts cooperative research projects focused on fish health issues throughout the region. These projects include controlled experiments in the Center’s wet lab as well as field-based studies. One long-term project, Klamath Fish Health Monitoring (2009 – present), examines the prevalence and severity of Ceratonova shasta and Parvicapsula minibicornis infection in both wild and hatchery juvenile Chinook salmon Klamath River out-migrants. The FHC use both qPCR and histology to determine infection status. In the spring, the FHC provides this information to Klamath River fishery managers to inform management actions to reduce disease effects on juveniles. Other applied research projects include the following issues:
- Effect of temperature and flow on salmon egg survival
- Effect of water quality on endangered Lost River sucker survival
- Mesocosm (5 years) and wetlab studies
- Monitoring of Ceratonova shasta in Feather River juvenile Chinook salmon
Project reports are available upon request.
Wild Fish Health Survey:
Since 1997, the CA-NV FHC has annually participated in the Wild Fish Health Survey (WFHS) by collecting fish tissue samples and performing laboratory tests for major fish pathogens in accordance with standardized procedures. These data sets are entered into a national database and is accessible to the public and resource managers. The CA-NV FHC works in cooperation with stakeholders such as other Federal, State, Tribal agencies and the aquaculture industry in surveying, sampling, and analyzing wild fish populations. The study and detection of pathogens is vital to prevent disease outbreaks and widespread pathogen distribution. Since the onset of the WFHS, the survey has dramatically increased our knowledge of the geographical range of known pathogens, expanded the documented number of host species, and allowed for detection of known viruses that had previously gone undetected.
Laboratory Data Management:
The USFWS fish health centers track their laboratory data using a common LIMS. LIMS is the acronym for a Laboratory Information Management System (LIMS). It is a system that allows laboratories to trace and track all samples or specimens that are received into the laboratory, tests performed; methods utilized, and their results through each step in the analysis process. Using a common data management system allows questions to be asked across the USFWS’s national fish health program.
qPCR:
Quantitative polymerase chain reaction, also known as qPCR, is a molecular tool being utilized by the Fish Health Center. This tool can detect small quantities of pathogen DNA by making millions of copies of DNA present in a sample. The accumulation of DNA is measured by equipment and software as the reaction progresses. This tool is very specific and sensitive and therefore can detect just a few molecules of initial DNA. The CA-NV FHC primarily uses qPCR for applied research projects.